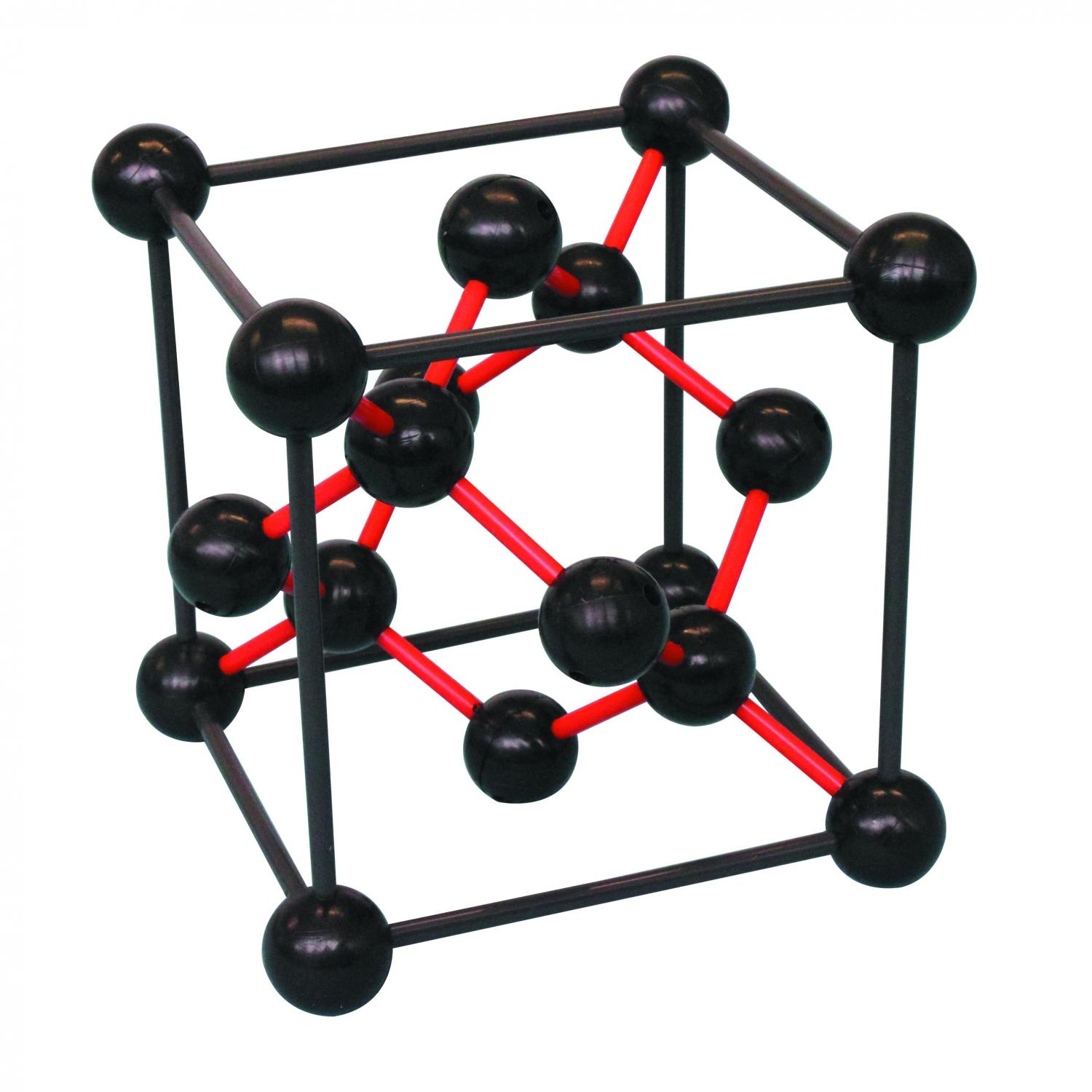
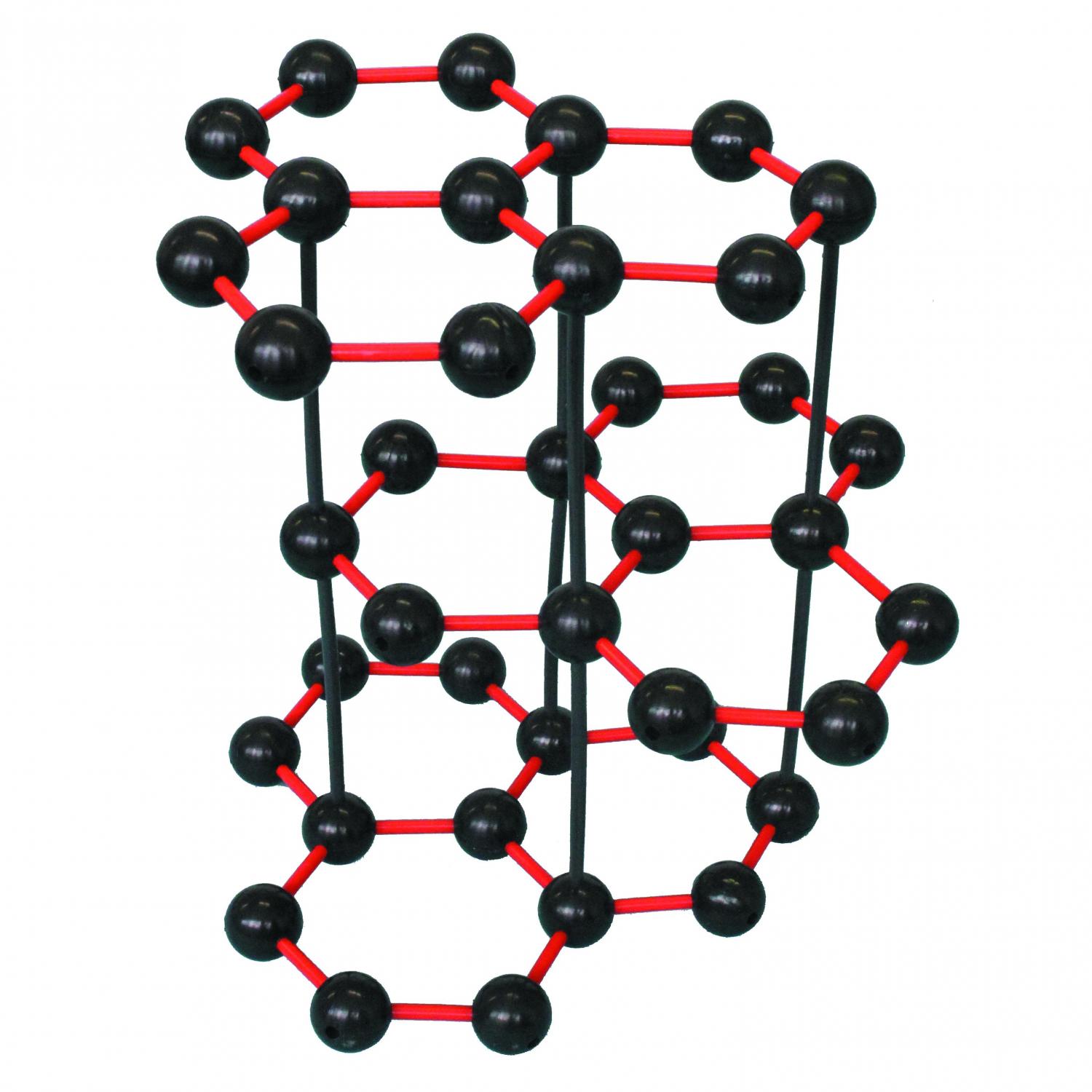
|
Ressources
|
-
Produits
- Data logging
- Measurement
- Observation
- Earth and Life Sciences
Forensics
Living organisms
Prepared slides
- Physics
- Chemistry
- Lab equipment
Glassware
Grounded glassware
Laboratory equipment
- Equipment
- Safety
- Data logging
- Catalogues
- Sales
- About us
- Contact